This article provides a brief overview of the role of ion channels in neuronal electrical signaling. Electrical signaling in the nervous system depends on the regulated movement of ions across cell membranes through specialized proteins known as ion channels. In Neuron, both Voltage-gated ion channels and Ligand-gated ion channels play crucial roles in generating and transmitting electrical signals. These processes lead to the formation of an action potential and enable communication between cells.
Excitable Cells and the Role of Ion Channels
Certain specialized cells in multicellular organisms can sense external stimuli and convert them into electrical signals, which they then transmit to other cells. These cells, known as excitable cells, are essential for many physiological processes such as nerve impulse transmission, muscle contraction, hormone release, sensory perception, and functions related to learning and memory. The ability of sensory cells, neurons, and muscle cells (myocytes) to generate electrical responses largely depends on Ion channel. These channels act as molecular gateways that regulate the movement of inorganic ions like Na⁺, K⁺, Ca²⁺, and Cl⁻ across the plasma membrane. By controlling ion flow in response to different stimuli, ion channels enable excitable cells to produce and transmit electrical signals.
Electrochemical Gradients and Ion Movement Across the Membrane
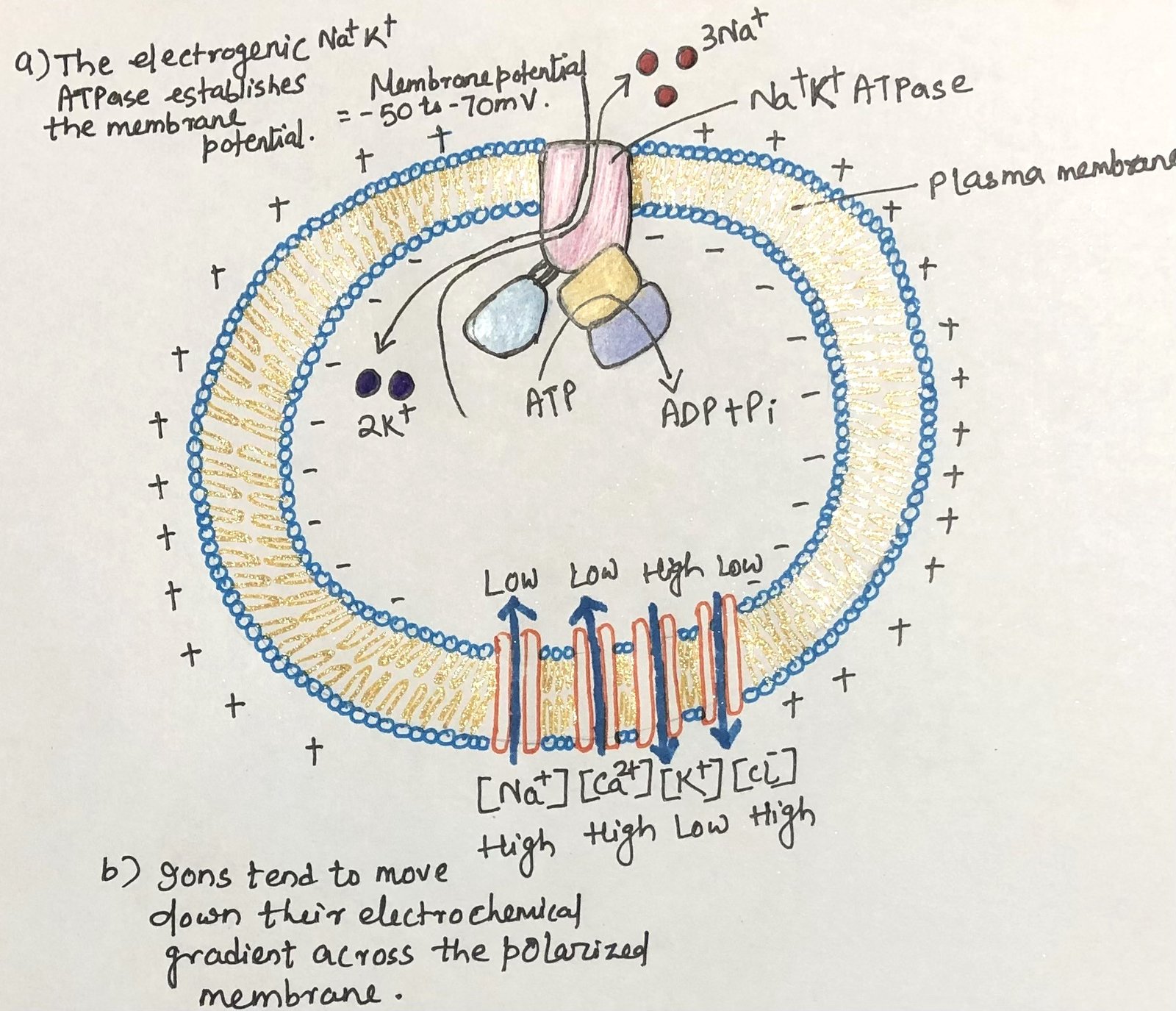
The Sodium–potassium pump plays a key role in maintaining electrical differences across the cell membrane. This pump actively moves three sodium ions (Na⁺) out of the cell while bringing two potassium ions (K⁺) into the cell, creating a slight electrical imbalance (Figure 1). As a result, the interior of the cell becomes more negatively charged compared with the exterior. Inside the cell, potassium ions are present in much higher concentrations, while sodium ions are relatively low compared with their levels outside the cell.
The direction in which ions move across a polarized membrane depends on their Electrochemical gradient. This gradient has two main components: the difference in ion concentration on the two sides of the membrane and the difference in electrical potential across the membrane, usually expressed as membrane potential (Vm). When the membrane potential is approximately–60 mV (with the inside negative), opening sodium or calcium channels allows these ions to flow spontaneously into the cell, causing membrane depolarization. In contrast, when potassium channels open, potassium ions move outward from the cell. Although this movement occurs against the electrical gradient, the strong concentration gradient drives the outward flow. For chloride ions (Cl⁻), the electrical potential of the membrane has a stronger influence; therefore, when chloride channels open, chloride ions tend to move outward across the membrane.
Ion Fluxes and Membrane Potential
Only a very small number of ions need to move across the membrane to create a meaningful change in membrane potential. Compared with the overall concentrations of ions such as Na⁺, K⁺, and Cl⁻ inside cells and in extracellular fluid, these movements are extremely minor. Therefore, the ion shifts that occur during electrical signaling in excitable cells do not significantly alter their overall concentrations. However, the situation is quite different for calcium ions. Since the intracellular level of Ca²⁺ is normally very low, even a small influx can cause a noticeable increase in cytosolic calcium levels, enabling it to function as an important Second messenger in various cellular processes.
At any moment, a cell’s membrane potential depends on the type and number of open ion channels. The controlled and timely opening and closing of Ion channel produce rapid, temporary changes in membrane potential. These fluctuations form the basis of electrical signaling, which allows the nervous system to coordinate vital activities such as muscle contraction, heartbeat regulation, and secretion from glandular cells.
Voltage-Gated Ion Channels in Neuronal Signaling
Communication within the nervous system occurs through interconnected networks of neurons, which transmit electrical signals known as action potentials. These impulses originate at the cell body and travel along a long extension called the axon. When the electrical signal reaches the end of the axon, it stimulates the release of a neurotransmitter at the synapse, thereby passing the signal to another neuron or a muscle cell.
This signaling process depends on three main types of Voltage-gated ion channels. Along the axon, voltage-gated sodium (Na⁺) channels (Figure 2) remain closed under resting conditions but open briefly when the membrane becomes locally depolarized, such as after stimulation by acetylcholine. This opening allows Na⁺ ions to rush into the cell, initiating depolarization. Shortly afterward, voltage-gated potassium (K⁺) channels open in response to this change, allowing K⁺ ions to flow out of the cell. This outward movement counteracts the earlier influx of sodium and restores the membrane toward its resting state, a process known as repolarization.
At the terminal end of the axon, voltage-gated calcium (Ca²⁺) channels play a crucial role. When the wave of depolarization reaches this region, these channels open and permit Ca²⁺ ions to enter the cell (step 1, figure 2). This influx of calcium triggers the release of neurotransmitters such as acetylcholine, while subsequent repolarization (step 2, figure 2) restores the membrane to its resting state after signal transmission.
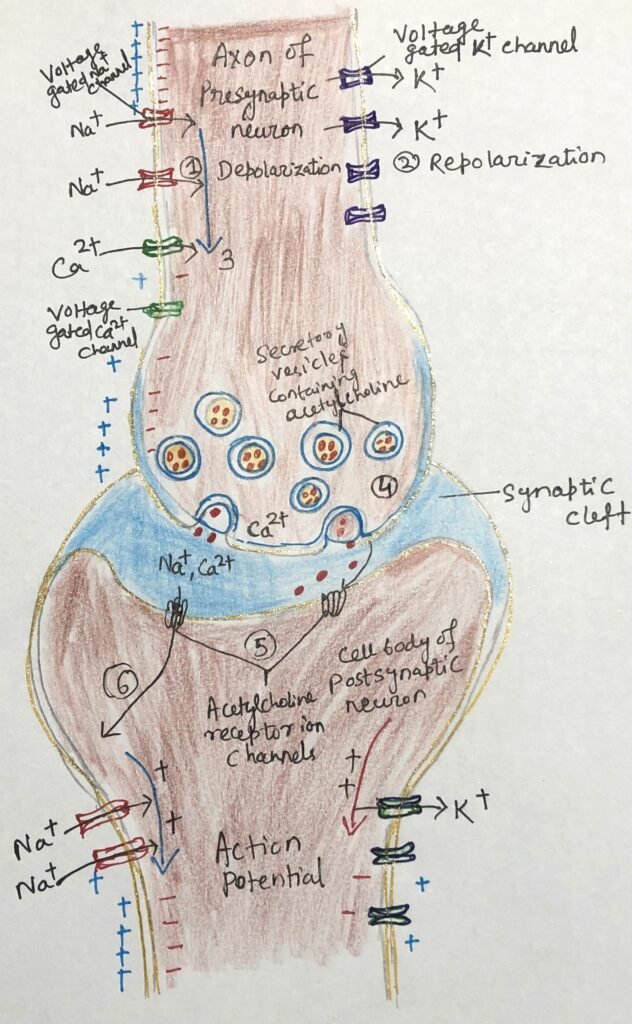
Function of Voltage-Gated Sodium and Potassium Channels
Voltage-gated Na⁺ channels show a high level of selectivity, allowing sodium ions to pass through much more readily than other positively charged ions. These channels open rapidly in response to changes in membrane potential but close again within milliseconds. The brief influx of Na⁺ ions causes localized depolarization, which in turn activates nearby potassium channels. The resulting efflux of K⁺ ions restores the negative charge inside the cell, thereby re-establishing the resting membrane potential.
Propagation of Action Potential and Synaptic Transmission
A short-lived wave of depolarization travels along the axon as each region of the membrane activates a nearby voltage-gated ion channel. When one segment depolarizes, it briefly opens adjacent sodium channels, followed quickly by potassium channels. After opening, each sodium channel enters a brief inactive state during which it cannot reopen. This ensures that the action potential moves in only one direction from the cell body toward the end of the axon.
As this wave reaches the axon terminal, it activates voltage-gated calcium channels, allowing Ca²⁺ ions to enter from the extracellular fluid (step 3, figure 2). The resulting increase in intracellular calcium concentration triggers the release of acetylcholine through exocytosis into the synaptic cleft (step 4, figure 2). The neurotransmitter then diffuses across the synapse and binds to receptors on the postsynaptic cell, causing depolarization and passing the signal forward in the neural circuit.
Gated ion channels transmit signals through two main mechanisms. They can alter the internal concentration of specific ions, enabling those ions to act as a second messenger, or they can change the membrane potential (Vm), thereby influencing other membrane proteins that respond to electrical changes. The transmission of an electrical impulse from one neuron to another demonstrates both of these signaling strategies.
Ligand-Gated Ion Channels and Synaptic Integration
Animal cells, particularly neurons, contain different types of ion channels that are regulated either by electrical signals, chemical signals, or both. Receptors that function directly as ion channels are known as Ionotropic receptor, distinguishing them from receptors that act through intracellular signaling pathways.
For example, Acetylcholine binds to an ionotropic receptor on the postsynaptic membrane. This receptor functions as a cation channel, allowing ions such as Na⁺, K⁺, and Ca²⁺ to pass through when activated. In contrast, neurotransmitters like glycine open channels that are selective for Cl⁻ ions.
The effect of neurotransmitter binding depends on the type of ion channel involved. The movement of ions across the membrane can either depolarize or hyperpolarize the target cell. Typically, a single neuron receives signals from many other neurons, each releasing different neurotransmitters with either excitatory or inhibitory effects. The overall membrane potential (Vm) of the neuron reflects the combined effect of all these inputs, and an action potential is generated only when the net depolarization reaches a sufficient threshold.
Receptor channels for neurotransmitters such as acetylcholine, glycine, glutamate, and Gamma-aminobutyric acid are activated by extracellular ligands. In addition, certain ion channels are regulated by intracellular molecules such as cAMP, cGMP, IP₃, Ca²⁺, and ATP, which act as second messengers. These mechanisms are particularly important in sensory processes like vision, smell, and taste.
Conclusion
Ion channels play a central role in the functioning of the nervous system by regulating the movement of ions across the cell membrane. Both Voltage-gated ion channels and ligand-gated ion channels work together to generate and transmit electrical signals in neurons. These coordinated activities lead to the formation of an action potential and enable communication between cells through neurotransmitters.
In addition, the integration of multiple signals and the involvement of intracellular molecules such as second messengers allow precise control of neuronal responses. Thus, ion channels are essential for complex processes such as sensation, muscle activity, and overall coordination within the nervous system.
You may also like:
- β-Adrenergic Receptor-Mediated Signal Transduction Involving Adenylyl Cyclase, cAMP, and PKA
- G Proteins at the Crossroads of Health and Disease

I, Swagatika Sahu (author of this website), have done my master’s in Biotechnology. I have around fourteen years of experience in writing and believe that writing is a great way to share knowledge. I hope the articles on the website will help users in enhancing their intellect in Biotechnology.