This article provides an overview of the role of protein kinases in controlling the cell cycle. Cell division is one of the most tightly regulated processes in biology, ensuring that new cells inherit an accurate copy of genetic information. This complex process is controlled by an intricate network of signaling pathways that coordinate growth, DNA replication, and division. Among the most important regulators are protein kinases, which act as molecular switches to drive the cell cycle forward at the correct time and in the proper sequence.
The Importance of Regulating Cell Division
In eukaryotic organisms, rapid cell division drives embryonic growth and tissue development. In contrast, most cells in the adult body enter a quiescent state and divide only when required for repair or renewal. When the mechanisms that restrict cell division fail, cells begin to proliferate uncontrollably, leading to severe consequences such as cancer.
To prevent this, the cell cycle is governed by a precisely ordered sequence of biochemical events that ensures each daughter cell receives a complete and functional set of cellular components. Central to this regulatory system are protein kinases, which act as molecular switches that control when a cell progresses through each stage of the cycle.
The Phases of the Eukaryotic Cell Cycle
In eukaryotic cells, division progresses through a series of clearly organized stages. During the S phase, the cell duplicates its DNA so that each future daughter cell will receive an identical genetic copy. This is followed by the G2 phase, a period of rapid growth in which the cell synthesizes essential proteins and increases in size in preparation for division.
The next stage is the M phase (mitosis). Here, the nuclear membrane surrounding the parent nucleus disassembles, and the duplicated chromosomes are separated and pulled toward opposite ends of the cell. New nuclear envelopes then form around each set of chromosomes. Finally, cytokinesis divides the cytoplasm, physically splitting the cell into two daughter cells.
After completing mitosis and the G1 phase, a cell faces a critical decision: it may continue into another round of division or exit the cycle and enter a resting state known as G0. Cells can remain in this quiescent phase for varying lengths of time, ranging from hours to the entire lifespan of the organism. If conditions later signal the need for division, cells in G0 re-enter the cycle by progressing back into the G1 phase.
Cyclin–CDK Control System
Protein kinases regulate cell division by phosphorylating key proteins at specific times, ensuring that the cell cycle progresses in an orderly manner. These enzymes function as cyclin–CDK complexes, composed of a regulatory cyclin and a catalytic cyclin-dependent kinase (CDK). CDKs alone are largely inactive, but cyclin binding activates the catalytic site and dramatically increases kinase activity. Different combinations of cyclins and CDKs operate at distinct stages of the cell cycle.
CDK activity is tightly controlled through four main mechanisms:
- Phosphorylation and dephosphorylation of CDKs
- Controlled degradation of cyclins
- Periodic synthesis of cyclins and CDKs
- Inhibition by specific CDK-inhibitory proteins
The cell sequentially activates and inactivates these complexes to act as a molecular clock and ensure that each phase of the cell cycle finishes before the next begins.
Regulation of CDKs by Phosphorylation
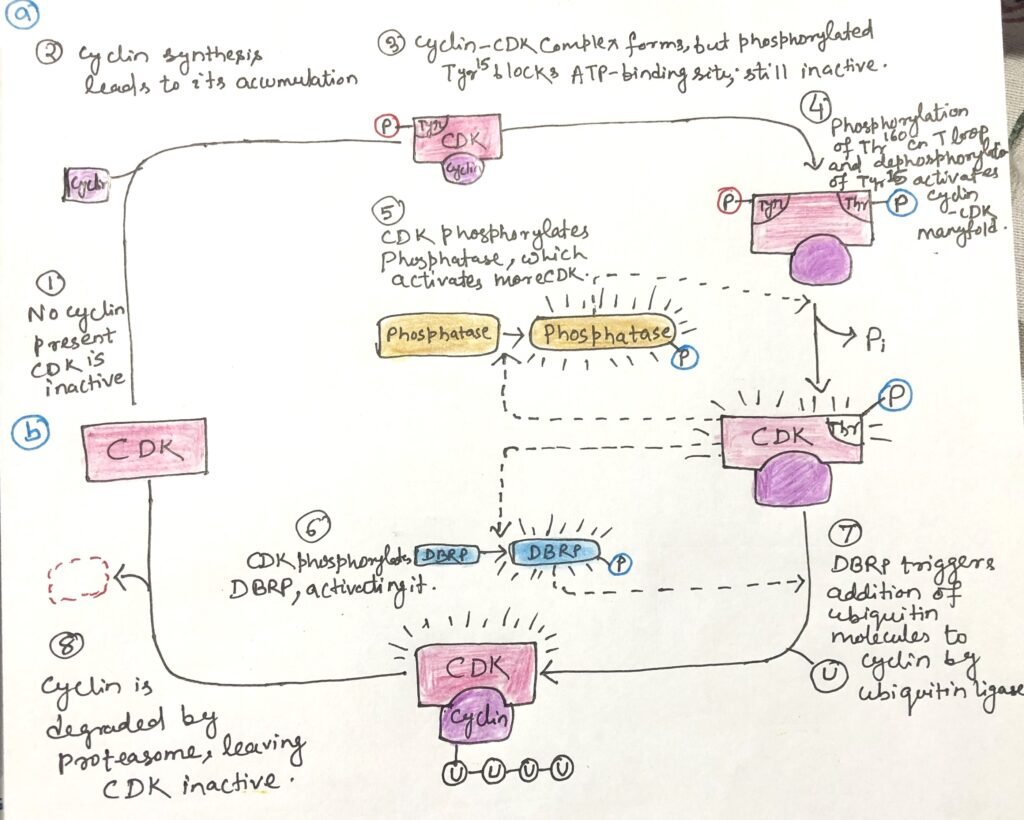
Phosphorylation at specific amino acid residues strongly regulates CDK activity. Phosphorylation of Thr160 in CDK2 stabilizes the enzyme in its active conformation by displacing the inhibitory T-loop away from the substrate-binding site, thereby allowing target proteins to bind. In contrast, removal of the phosphate group from Tyr15 eliminates a negative charge that otherwise blocks ATP from entering its binding pocket.
This activation process reinforces itself because CDK-mediated phosphorylation activates the phosphatase that removes the inhibitory phosphate. Together, these events greatly enhance CDK activity and enable the phosphorylation of downstream proteins required for cell-cycle progression (Figure 1a).
A single-strand break in DNA can halt the cell cycle at the G2 stage. This damage activates checkpoint proteins that initiate a signaling cascade, ultimately blocking the enzyme responsible for removing the inhibitory phosphate from Tyr15 on CDK. As a result, the CDK remains inactive, and the cell cannot enter the mitotic phase. After the cell repairs damaged DNA, it resumes division and lifts the checkpoint signals.
Cyclin Degradation and Mitotic Progression
Timely destruction of mitotic cyclins is essential for controlling CDK activity during the cell cycle (Figure 1b). Entry into and progression through mitosis require the activation and subsequent breakdown of cyclins A and B, which stimulate the M-phase CDK. These cyclins carry a short amino-terminal sequence called the destruction box, which marks them for degradation.
A destruction-box–recognizing protein identifies this sequence and promotes attachment of ubiquitin to the cyclin through the action of ubiquitin ligase. The addition of additional ubiquitin molecules forms a chain that serves as a signal for the proteasome, which then breaks down the cyclin. This regulated proteolysis ensures proper timing of mitotic events and allows the cell cycle to proceed to the next stage.
Feedback Control of Cyclin Levels
Cyclin destruction is tightly linked to CDK activity through a feedback loop. Newly synthesized cyclin binds to and activates CDK, which then phosphorylates and activates the destruction-box–recognizing protein (DBRP). Active DBRP promotes cyclin degradation, causing cyclin levels to fall and CDK activity to decline. As CDK activity drops, dephosphorylation gradually inactivates DBRP. This allows cyclin levels to rise again through new synthesis.
Beyond cell-cycle regulation, the ubiquitin–proteasome system plays a broader role in the continuous turnover of cellular proteins, a process essential for maintaining normal cellular function.
Regulation Through Cyclin and CDK Synthesis
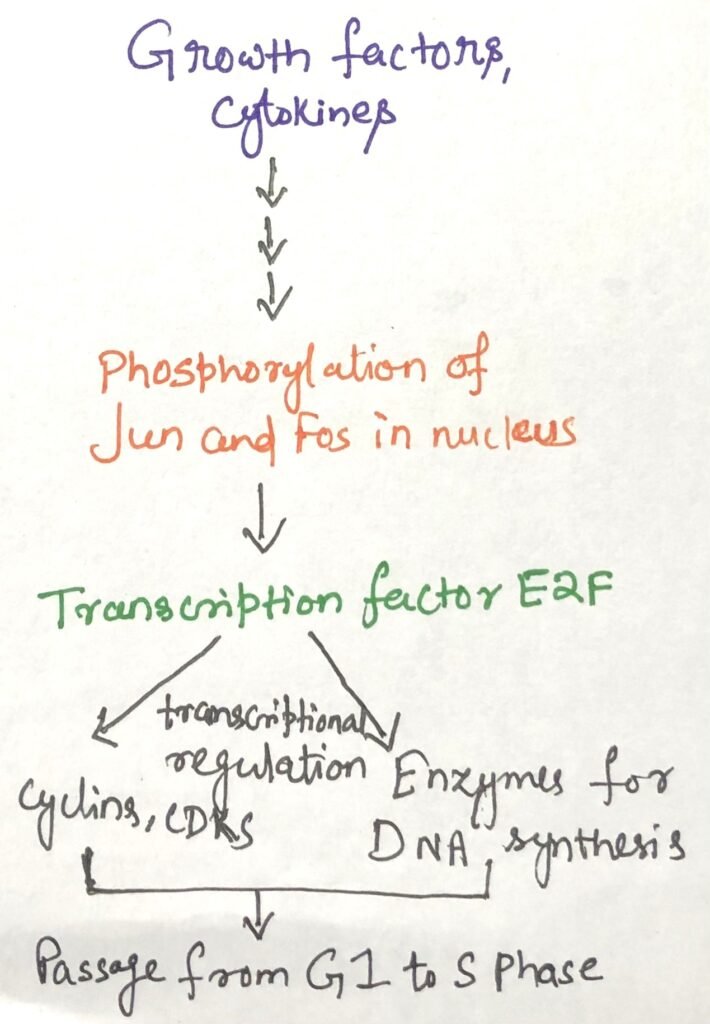
Regulation of the rate at which cells produce cyclins and CDKs also controls CDK activity. External signals such as growth factors and cytokines activate transcription factors like Jun and Fos, which stimulate the expression of genes required for cell-cycle progression. These genes include those encoding cyclins, CDKs, and the transcription factor E2F.
E2F further promotes the production of enzymes needed for DNA and deoxynucleotide synthesis, enabling the cell to transition into the S phase and begin DNA replication (Figure 2).
CDK Inhibitors and Conserved Regulation
Specific inhibitory proteins can bind to CDKs and block their activity, providing another layer of control over the cell cycle. Together, the four regulatory mechanisms determine whether a cell proceeds with division, enters a resting state, differentiates, or resumes proliferation after a period of quiescence.
Although the number and combinations of cyclins and CDKs vary among species, the fundamental strategy for controlling the cell cycle has remained highly conserved throughout the evolution of eukaryotic cells.
Key Cellular Targets of CDKs
The range of proteins regulated by CDKs continues to expand, but several well-studied examples reveal how these kinases coordinate major structural changes during cell division.
One important target is lamin, a protein that forms a supportive network beneath the nuclear envelope. As a cell prepares for mitosis, CDK-mediated phosphorylation of lamin causes this network to disassemble, leading to the breakdown of the nuclear envelope so chromosomes can separate properly.
Another major target is the actomyosin contractile system responsible for splitting the cell during cytokinesis. After division is completed, a CDK phosphorylates a regulatory subunit of myosin, detaches myosin from actin filaments, and shuts down the contractile machinery. Later, the removal of this phosphate group allows the contractile system to reassemble for the next cycle of cell division.
pRb–E2F Pathway and DNA Damage Checkpoint
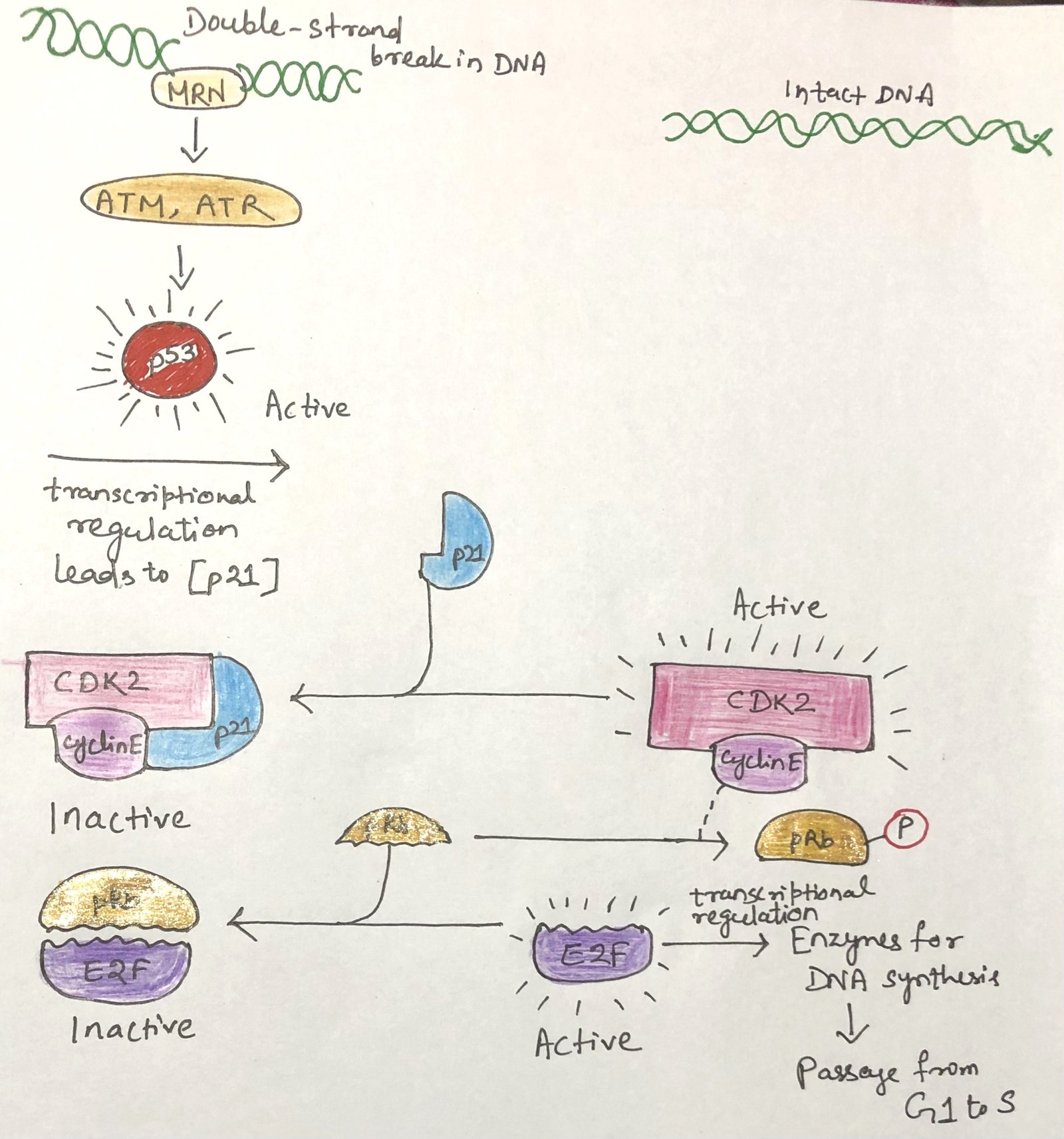
A major target of CDKs is the retinoblastoma protein (pRb), a key regulator of the transition from G1 to S phase. In its unphosphorylated form, pRb binds the transcription factor E2F, preventing it from activating genes required for DNA synthesis. As long as E2F remains inhibited, the cell cannot move from G1 into S phase (Figure 3). When signals promoting cell division are present, the cyclin E–CDK2 complex phosphorylates pRb, releasing E2F and allowing the transcription of genes needed for DNA replication.
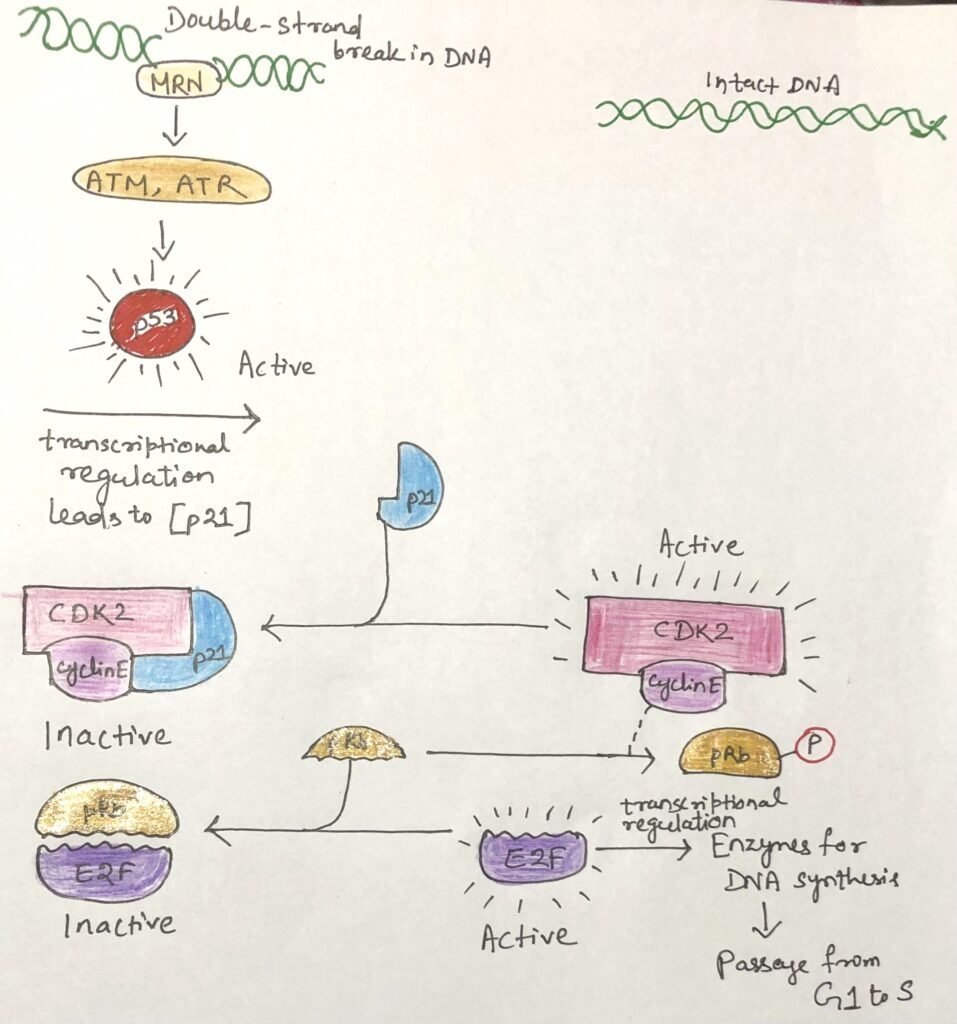
DNA damage activates an additional protective pathway. The kinases ATM and ATR detect damaged DNA and phosphorylate p53, turning it into an active transcription factor. Activated p53 stimulates production of p21, a CDK inhibitor that blocks cyclin E–CDK2 activity. As a result, pRb stays unphosphorylated and continues to suppress E2F, arresting the cell cycle in G1 and giving the cell time to repair its DNA.
If the damage is too severe to repair, the pathway triggers apoptosis, eliminates the damaged cell, and prevents the spread of harmful mutations that could lead to cancer.
Conclusion
Protein kinases, particularly cyclin–CDK complexes, form the central control system that governs progression through the eukaryotic cell cycle. Through phosphorylation, regulated protein synthesis, targeted proteolysis, and inhibitory pathways, these kinases ensure that each stage of division occurs in the correct order and only under favorable conditions.
This highly coordinated network not only preserves genomic stability but also protects organisms from uncontrolled cell proliferation. Understanding these regulatory mechanisms remains essential for advancing research in cancer biology, molecular medicine, and targeted therapeutic development.
You may also like:

I, Swagatika Sahu (author of this website), have done my master’s in Biotechnology. I have around fourteen years of experience in writing and believe that writing is a great way to share knowledge. I hope the articles on the website will help users in enhancing their intellect in Biotechnology.