This article describes the diverse roles of mitochondria beyond ATP production. Mitochondria are often introduced as the powerhouses of the cell, yet their functions extend far beyond ATP production. These dynamic organelles participate in specialized processes that help organisms adapt, survive, and maintain internal balance. From generating heat and synthesizing steroid hormones to directing programmed cell death, mitochondria play central roles in diverse physiological pathways.
Mitochondria as Multifunctional Regulators of Cellular Physiology
Mitochondria are widely recognized as the primary generators of cellular ATP, yet their biological significance extends far beyond energy production. These dynamic organelles adapt their functions to meet the specialized demands of different tissues and physiological conditions. In brown and beige adipose tissue, mitochondria act as cellular heat generators, dissipating energy as heat to maintain body temperature during cold exposure. In steroid-producing organs such as the adrenal cortex and the gonads, they provide the essential biochemical environment for the synthesis of steroid hormones. Across nearly all cell types, mitochondria also serve as central regulators of programmed cell death, helping determine whether a cell survives or undergoes apoptosis. Through their roles in metabolism, signaling, and cellular fate, mitochondria emerge not merely as powerhouses but as versatile coordinators of vital physiological processes.
Mitochondrial Thermogenesis in Brown Adipose Tissue
When cellular ATP levels are sufficient, the rate of respiratory activity naturally declines. However, in certain physiological situations, mitochondria redirect energy metabolism away from ATP synthesis toward heat production. Newborn mammals, including humans, possess a specialized fat tissue known as brown adipose tissue, whose primary role is to generate heat rather than store energy. This tissue appears brown due to its exceptionally high mitochondrial density and the abundance of cytochromes containing heme groups that strongly absorb visible light.
Mitochondria in brown fat cells resemble those found in other tissues but contain a distinctive inner-membrane protein called uncoupling protein 1. This protein creates an alternative route for protons to flow back into the mitochondrial matrix without passing through ATP synthase. By bypassing ATP formation, the proton gradient is dissipated as thermal energy, effectively converting the energy of fuel oxidation into heat that helps maintain body temperature.
The thermogenic function of brown adipose tissue is not limited to newborns. Hibernating animals also rely heavily on this mechanism, using uncoupled mitochondrial respiration to produce heat and survive extended periods of cold and reduced metabolic activity.
Mitochondrial Role in Steroid Hormone Biosynthesis
Mitochondria serve as major centers for the production of steroid hormones, including sex hormones, glucocorticoids, mineralocorticoids, and the hormonally active form of vitamin D. Enzymes of the cytochrome P-450 family catalyze a series of hydroxylation reactions that derive these molecules from cholesterol or closely related sterols. Each of these enzymes contains an essential heme group that enables oxygen activation.
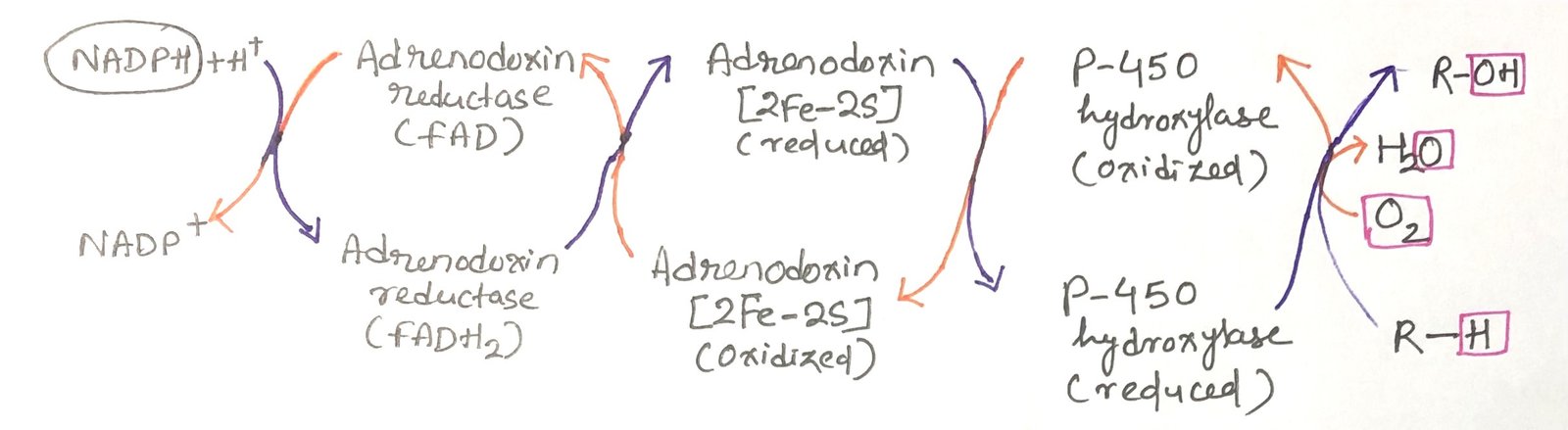
During these hydroxylation reactions, the process incorporates one atom of molecular oxygen into the substrate and reduces the second oxygen atom to water. Because of this dual function, cytochrome P-450 enzymes are classified as monooxygenases. The overall reaction can be summarized as
R-H + O₂ + NADPH + H⁺ → R-OH + H₂O + NADP⁺
In this process, both NADPH and the substrate undergo oxidation.
Steroid-producing cells contain abundant mitochondria specifically adapted for hormone synthesis. These mitochondria are typically larger than those found in other tissues and possess highly folded inner membranes that increase the surface area available for enzymatic reactions. The inner mitochondrial membrane embeds numerous cytochrome P-450 enzymes and orients their catalytic sites toward the matrix.
Electron transfer within the mitochondrial P-450 system follows a multi-step pathway. Electrons originating from NADPH are first passed to a flavoprotein and then to an iron–sulfur protein before finally reaching the P-450 heme center (Figure 1). This electron transport chain enables oxygen activation and substrate hydroxylation. Although many different P-450 enzymes exist, they all share the common feature of a heme group that binds oxygen and a substrate-binding pocket that determines reaction specificity.
Endoplasmic Reticulum Cytochrome P450 Enzymes in Detoxification and Drug Metabolism
A second major group of cytochrome P450 enzymes is located in the endoplasmic reticulum of liver cells. While they perform reactions similar to mitochondrial P450 enzymes, their main role is the breakdown of a wide range of hydrophobic molecules, many of which are xenobiotics (chemicals that do not naturally occur in the body and are often produced industrially).
These liver enzymes have broad and overlapping substrate specificities, enabling them to act on a wide range of compounds. By adding hydroxyl groups to hydrophobic substances, they make these molecules more water-soluble. This chemical modification enables the kidneys to remove them from the bloodstream and eliminate them through urine. Many commonly prescribed medications are among the compounds that these enzymes process.
The activity of cytochrome P450 enzymes strongly influences how long a drug remains active in the bloodstream. Thus, it affects its therapeutic effectiveness. Individuals differ in their genetic makeup of these enzymes and in the extent to which lifestyle factors, such as long-term alcohol consumption, induce certain P450 enzymes. As a result, both genetics and personal history can play an important role in determining the appropriate dosage of medications for an individual.
Mitochondrial Control of Apoptosis
Programmed cell death, or apoptosis, is a carefully regulated process in which individual cells are eliminated for the overall benefit of the organism. It plays an essential role in normal development, tissue maintenance, and the recycling of cellular components. Apoptosis can begin through signals from outside the cell, such as those received by membrane receptors, or through internal disturbances, including DNA damage, viral infection, accumulation of reactive oxygen species, or exposure to heat and other cellular stresses.
Mitochondria are central to the initiation of this process. In response to death signals, the outer mitochondrial membrane becomes more permeable, allowing cytochrome c to move from the intermembrane space into the cytosol. This change in membrane permeability occurs through the opening of the permeability transition pore complex. This complex is a multi-protein structure, and factors regulating its activity either promote or inhibit apoptosis (Figure 2).
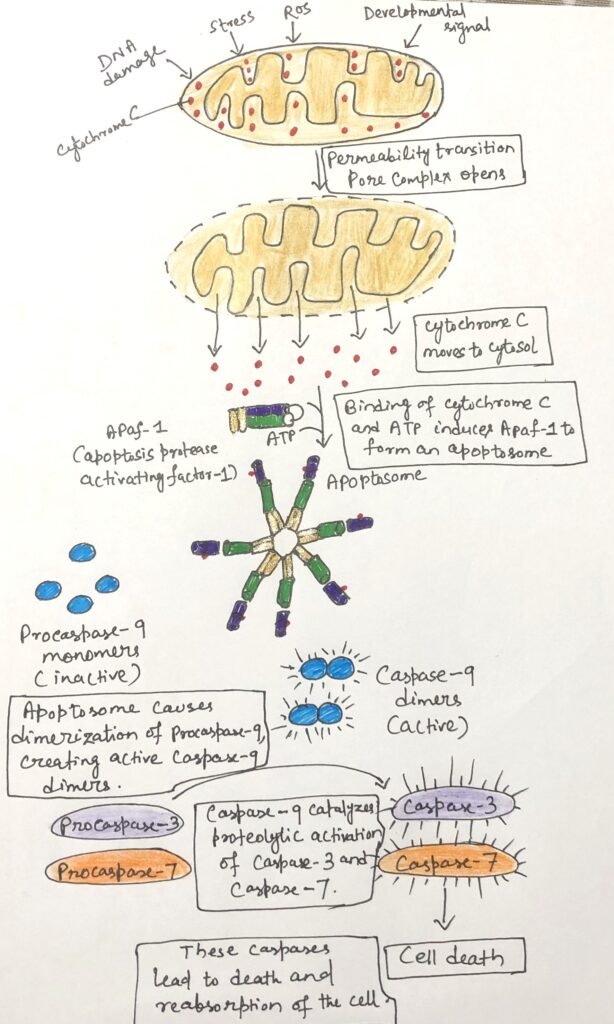
Once in the cytosol, cytochrome c binds to molecules of Apaf-1, triggering the assembly of a large protein complex known as the apoptosome. This complex acts as a platform for the activation of procaspase-9 into its active form, caspase-9. A key cysteine residue in the active site characterizes caspases, a family of highly specific proteases. They cleave proteins at sites located after aspartate residues. Activated caspase-9 then initiates a cascade of proteolytic events, in which one caspase activates another, ultimately leading to the systematic breakdown of cellular components and the orderly dismantling of the cell.
Conclusion
Mitochondria are far more than simple energy-producing organelles. Their ability to switch between ATP generation, heat production, steroid hormone biosynthesis, detoxification support, and the regulation of programmed cell death highlights their remarkable functional versatility. By integrating metabolic activity with cellular signaling and survival pathways, mitochondria influence processes ranging from temperature regulation and hormone balance to drug metabolism and cell fate decisions. Understanding these diverse roles not only deepens our knowledge of cellular physiology but also provides important insights into metabolic disorders, endocrine diseases, and therapeutic strategies targeting mitochondrial function.
You may also like:
- Mitochondrial DNA Origin and Mutational Effects
- Tumor Immune Escape Mechanisms and Apoptotic Resistance

I, Swagatika Sahu (author of this website), have done my master’s in Biotechnology. I have around fourteen years of experience in writing and believe that writing is a great way to share knowledge. I hope the articles on the website will help users in enhancing their intellect in Biotechnology.