This article provides a brief description of coliform bacteria as indicators of water quality and outlines the standard bacteriological techniques used for their detection and enumeration. Water intended for human consumption must meet strict microbiological standards to ensure safety and prevent disease transmission. Coliform bacteria serve as important indicator organisms for evaluating fecal contamination in water supplies. This article explores the biological significance of coliforms and the standard bacteriological methods used for their detection and assessment.
Microbial Indicators of Fecal Contamination
Water bodies frequently become contaminated through the discharge of human and animal waste. Enteric pathogens that cause diseases such as typhoid, dysentery, and gastroenteritis reach aquatic systems primarily via fecal matter. Because direct detection of all pathogens in water is technically complex, time-consuming, and often impractical, microbiologists rely on indicator organisms to assess sanitary quality.
Among these indicators, Escherichia coli and other members of the coliform group, along with fecal streptococci (e.g., Streptococcus faecalis) and Clostridium perfringens, are of particular importance. These bacteria are normal inhabitants of the intestinal tract of humans and warm-blooded animals and are consistently excreted in feces. Their detection in water, therefore, serves as strong evidence of fecal contamination. The presence of coliform bacteria does not necessarily confirm the existence of specific pathogens. However, it signals that conditions allow intestinal microorganisms, including potential disease-causing agents, to enter the water supply. For this reason, coliform testing has become a cornerstone of bacteriological water quality assessment and public health surveillance.
Characteristics and Biochemical Differentiation of Coliform Bacteria
The coliform group comprises aerobic and facultatively anaerobic, Gram-negative, non-spore-forming rod-shaped bacteria capable of fermenting lactose with the production of acid and gas within 48 hours at 35–37°C. Traditionally, the principal representatives of this group include Escherichia coli and Enterobacter aerogenes (now reclassified as Klebsiella aerogenes).
Coliform bacteria belong to the broader enteric group, which also includes genera such as Salmonella, Shigella, Klebsiella, Proteus, and Serratia. These organisms share striking similarities in morphology and cultural characteristics, making differentiation based solely on microscopic appearance and colony morphology unreliable. Therefore, biochemical testing becomes essential for accurate identification.
IMViC Reactions for Differentiation
A series of four biochemical tests, collectively known as the IMViC reactions, is widely employed to distinguish E. coli from Enterobacter species and other coliforms. The acronym IMViC represents:
- Indole Test (I)
Determines the ability to degrade tryptophan to indole.- E. coli → Indole positive
- Enterobacter aerogenes → Indole negative
- Methyl Red Test (M)
Detects stable acid production from glucose fermentation.- E. coli produces a large quantity of stable acids, lowering the pH sufficiently to turn the methyl red indicator red (positive result).
- Enterobacter aerogenes produces less acid, resulting in a negative reaction.
- Voges–Proskauer Test (Vi)
Identifies the production of acetoin (acetylmethylcarbinol) from glucose metabolism.- E. coli → Negative
- Enterobacter aerogenes → Positive
- Citrate Utilization Test (C)
Assesses the ability to use sodium citrate as the sole carbon source.- Enterobacter aerogenes grows in citrate medium (positive).
- E. coli does not grow under these conditions (negative).
Together, the IMViC pattern typically observed is:
- E. coli → ++––
- Enterobacter aerogenes → ––++
These biochemical reactions form the foundation of laboratory differentiation of coliform bacteria and play a crucial role in bacteriological water quality analysis.
Standard Guidelines for Bacteriological Examination of Water
The procedures for microbiological analysis of water and wastewater are comprehensively described in Standard Methods for the Examination of Water and Wastewater, jointly developed by the American Public Health Association, the American Water Works Association, and the Water Environment Federation (formerly the Water Pollution Control Federation).
Additionally, the United States Environmental Protection Agency has issued detailed protocols under the title Microbiological Methods for Monitoring the Environment: Water and Wastewater, which provide standardized procedures for detecting indicator organisms, including coliform bacteria.
Accurate bacteriological analysis depends not only on laboratory techniques but also on proper sample collection and handling. Failure to maintain sampling integrity may lead to misleading results. Therefore, strict adherence to the following principles is essential.
Essential Requirements for Water Sampling
- Use of Sterile Containers
Samples must be collected in sterile, properly sealed bottles to prevent external contamination. - Representative Sampling
The collected sample should accurately reflect the microbiological quality of the source water supply. - Prevention of Secondary Contamination
Care must be taken during and after sampling to avoid introducing extraneous microorganisms. - Prompt Examination
Microbiological testing should be performed as soon as possible after collection to prevent changes in the bacterial population. - Proper Storage Conditions
If immediate analysis is not feasible, samples should be refrigerated at temperatures between 0°C and 10°C to minimize bacterial growth or die-off before testing.
Standard Plate Count (SPC) in Water Quality Assessment
The standard plate count (SPC) involves plating measured aliquots of a water sample onto appropriate nutrient media and incubating them to allow viable microorganisms to form visible colonies. Each colony theoretically represents a colony-forming unit (CFU) originating from a single viable cell or a cluster of cells.
Interpretation of plate count results requires careful consideration. The mere number of bacteria present does not necessarily correlate with health risk, as a small number of pathogenic organisms may pose greater danger than a large population of harmless saprophytic bacteria. Therefore, the standard plate count serves primarily as an indicator of overall microbial load rather than a direct measure of pathogenic contamination (Figure 1).
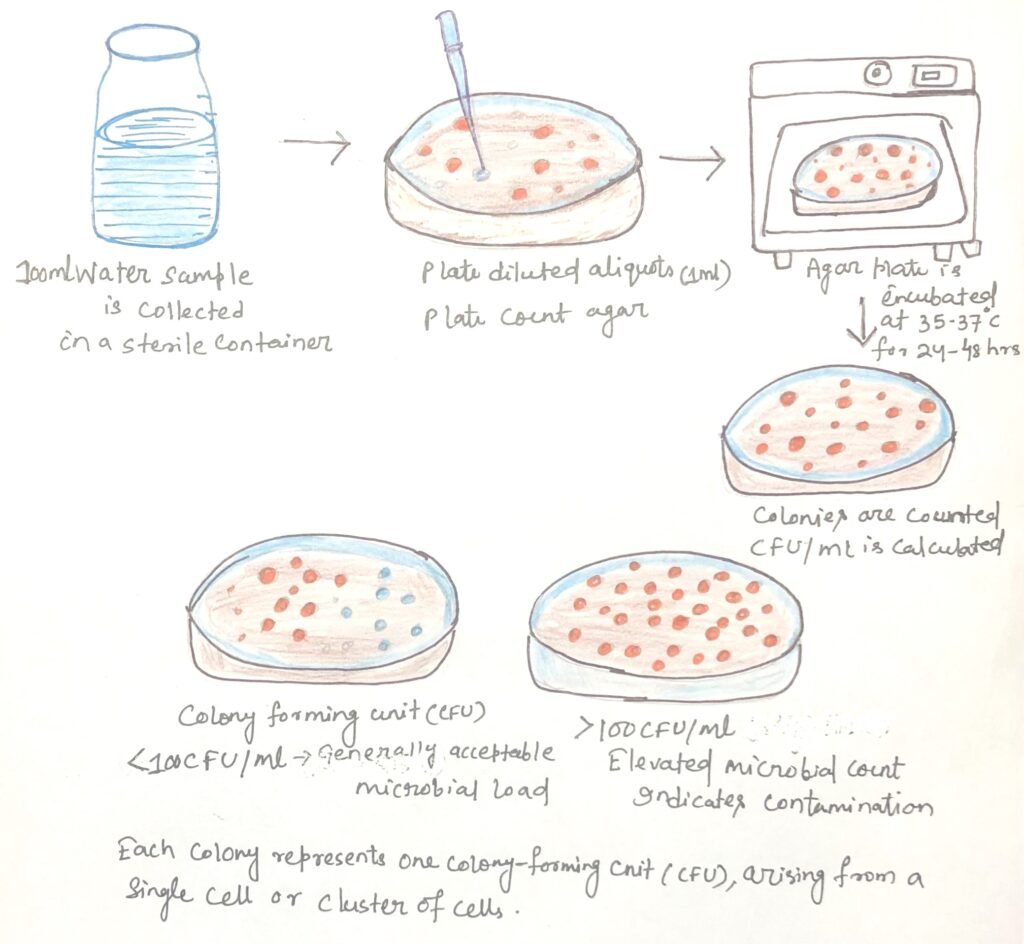
In potable water supplies, high-quality water typically yields low bacterial counts, generally fewer than 100 colony-forming units per milliliter (CFU/mL), depending on regulatory standards. The SPC is particularly valuable in evaluating the efficiency of water treatment processes such as sedimentation, filtration, and chlorination. Comparing microbial counts before and after treatment allows quantification of microbial reduction and evaluation of treatment efficiency.
Selective Media and Systematic Detection of Coliform Bacteria
The use of selective and differential culture media enables the detection of coliform organisms in water by inhibiting non-target bacteria and allowing characteristic reactions to develop. Traditional microbiological analysis for coliforms generally follows three sequential steps:
- Presumptive Test
- Confirmed Test
- Completed Test
Together, these steps increase accuracy and ensure reliable identification of coliform contamination.
Membrane-Filter Technique For Coliform Detection
Microbiologists use the membrane filtration (MF) method as an efficient and widely accepted technique for examining potable water with low bacterial density. This method allows quantitative estimation of coliform organisms.
Principle
The method passes a measured volume of water through a sterile membrane filter (0.45 µm pore size). The filter retains bacteria from the sample and is subsequently incubated on selective medium to permit colony formation.
Procedure
- A sterile membrane filter disk is aseptically placed in a filtration apparatus.
- A known volume of the water sample is drawn through the membrane under vacuum; microorganisms are trapped on its surface.
- The membrane is carefully removed using sterile forceps and transferred onto:
- An absorbent pad saturated with a selective growth medium,
- The surface of a selective and differential agar medium in a Petri dish.
- The prepared plates need incubation under appropriate temperature conditions (commonly 35–37°C for total coliforms).
- After incubation, colonies develop on the membrane at sites where bacteria were retained during filtration.
Interpretation
Characteristic colony morphology and color changes (depending on the selective medium used) allow identification and enumeration of coliform bacteria, expressed as colony-forming units per 100 mL (CFU/100 mL).
The membrane filtration method is especially advantageous because it:
- Provides quantitative results
- Allows examination of large sample volumes
- Is more rapid than traditional multiple-tube fermentation methods
Most Probable Number (MPN) Method for Coliform Detection
The Most Probable Number (MPN) method is also known as the multiple-tube fermentation technique. It is a statistical procedure that estimates the concentration of coliform bacteria in water samples. This method is particularly useful when testing water with low bacterial density or when the sample contains particulate matter that may interfere with membrane filtration.
Principle
The MPN technique is based on the probability of detecting coliform organisms through lactose fermentation with the production of acid and gas. Laboratory personnel inoculate serial dilutions of the water sample into lactose broth tubes fitted with inverted Durham tubes to detect gas formation. After incubation, they identify tubes showing acid and gas production as presumptive positive for coliforms.
Procedure
- Presumptive Test – Water samples are inoculated into lactose broth tubes and incubated. Gas formation indicates possible coliform presence.
- Confirmed Test – Samples from positive tubes are transferred to selective media to verify coliform growth.
- Completed Test – Further subculturing and biochemical tests confirm the identity of coliform bacteria.
Microbiologists compare the number of positive tubes at each dilution level with standard MPN tables to estimate the bacterial concentration, usually expressed as MPN per 100 mL of water.
Although slower than the membrane filtration method, the MPN technique remains a widely accepted and reliable approach for bacteriological examination of water, particularly in routine quality monitoring.
Regulatory Standards for Coliform in Drinking Water
Regulatory agencies worldwide establish strict microbiological criteria to ensure the safety of drinking water. According to guidelines issued by the World Health Organization (WHO) and the United States Environmental Protection Agency (EPA), potable water must contain no detectable coliform bacteria in 100 mL of sample.
The presence of total coliforms indicates possible system vulnerability or contamination, whereas the detection of Escherichia coli specifically confirms recent fecal contamination and represents an immediate public health concern. Many national standards, therefore, require zero tolerance for E. coli in treated drinking water supplies.
Routine monitoring, compliance testing, and corrective actions are mandatory components of public water system management to prevent waterborne disease outbreaks.
Conclusion
Coliform bacteria serve as reliable microbial indicators of water quality. They play a central role in assessing fecal contamination in drinking water supplies. Direct testing for all waterborne pathogens is difficult and time-consuming. Therefore, indicator organisms such as Escherichia coli are used for routine monitoring. They provide a practical and reliable alternative.
Through established techniques, including the Standard Plate Count, Membrane Filtration method, and the Most Probable Number (MPN) procedure, microbiologists can effectively evaluate the sanitary status of water systems. Adherence to regulatory standards and systematic surveillance ensures early detection of contamination, thereby safeguarding public health and preventing waterborne disease outbreaks.
You may also like:
- Bacterial Exotoxins and Their Pathogenic Effects
- Endotoxins and Associated Virulence Determinants in Bacterial Pathogenesis

I, Swagatika Sahu (author of this website), have done my master’s in Biotechnology. I have around fourteen years of experience in writing and believe that writing is a great way to share knowledge. I hope the articles on the website will help users in enhancing their intellect in Biotechnology.

